
Every gas stream is unique.
Each gas has a different tendency to be dissolved in lubricating oil. All factors determining this tendency must be taken into account, including the actual operating conditions:
1. Pressure
The higher the compression, the higher the tendency of the gas to be dissolved in the oil.
2. Temperature
The higher the compression temperature, the lower the tendency of the gas to be dissolved in the oil.
3. Molecular
weight The higher the molecular weight of the gas, the higher its tendency to be dissolved in the oil. “Heavy ” hydrocarbons such as toluene are more easily dissolved than “light” ones such as methane or propane.
4. Polarity
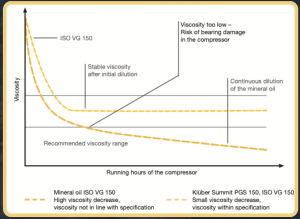
When it comes to polarity, the principle of “likeness ” applies. Polar gases such as ammonia are easily dissolved in polar oils such as polyglycol, while non-polar gases such as hydrocarbons are less easily dissolved in polar oils. Taking these criteria into account, the tendency of a gas to be dissolved in lubricating oil can be calculated before the rotary screw compressor is filled with lubricant. This provides valuable information about oil viscosity under actual operating conditions and helps ensure that the lubricant meets the manufacturer ’ s viscosity requirements in real-life situations.